Edgewise Announces Positive Long-Term Sevasemten Data Demonstrating Sustained Functional Stabilization in Becker Muscular Dystrophy Through 3.5 Years of Follow-Up
PR Newswire
BOULDER, Colo., March 10, 2026
–The MESA data show stabilization of function in participants on sevasemten versus the predicted functional decline observed in Becker natural history studies –
– Data reinforce prior clinical findings of sevasemten in Becker, a rare disease with no approved treatments –
–The GRAND CANYON placebo-controlled pivotal cohort is on track to readout in 4Q 2026 –
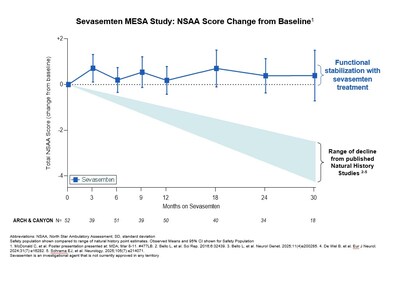
BOULDER, Colo., March 10, 2026 /PRNewswire/ -- Edgewise Therapeutics, Inc. (Nasdaq: EWTX), a leading muscle disease biopharmaceutical company, today presented at the 2026 MDA Clinical and Scientific Conference, long-term data from its MESA open-label extension study of sevasemten in Becker muscular dystrophy (Becker), a devastating genetic disorder with no approved therapeutic interventions. The data show participants on sevasemten experienced stabilization of function over 3.5 years in marked contrast to the functional decline expected from Becker natural history data.
"For the first time with an investigational agent, we are seeing long-term data where individuals with Becker are stable in the face of a disease that we have seen would otherwise lead to a significant functional decline," said Joanne Donovan, M.D., Ph.D., Chief Medical Officer of Edgewise Therapeutics. "We remain on track to deliver pivotal GRAND CANYON data for sevasemten and advancing towards the first marketing application for the treatment of Becker."
MESA is an open-label extension evaluating sevasemten long-term safety, tolerability, and efficacy in adults and adolescents with Becker previously treated in sevasemten clinical trials. Nearly all eligible participants (99%) from prior sevasemten studies (including ARCH, CANYON/GRAND CANYON, and DUNE) chose to enroll in the MESA open-label extension study.
Key findings reported after 12 months of ARCH and CANYON participation and further follow-ups in MESA:
- Sustained stabilization over 3.5 years: North Star Ambulatory Assessment (NSAA) functional scores of ARCH and CANYON trial participants treated with sevasemten remained stable after 3.5 years and 2 years, respectively. The CANYON participants on placebo who rolled into the MESA study also had NSAA functional scores that trended upward during the first year after switching to sevasemten.
- Divergence from Becker natural history: Once Becker functional decline begins, its course typically continues along that downward trajectory. Multiple natural history studies in Becker report NSAA scores declining by an average of 1.0 to 1.7 points annually; this equates to an expected average functional decline of 3.0 to 5.1 points over 3 years.1-4
- In MESA, the NSAA scores observed in the sevasemten treatment arm diverged markedly from predicted natural history declines:
- CANYON over 2 years: +0.1 improvement (treated) vs. -2.9 decline (predicted natural history)
- ARCH over 3.5 years: +0.1 improvement (treated) vs. -5.3 decline (predicted natural history)
Sevasemten, an investigational fast skeletal myosin inhibitor designed to protect muscle from contraction-induced damage, continued to show a favorable safety profile in MESA with long-term exposure for 3+ years.
The Company expects to announce top-line results of the sevasemten placebo-controlled pivotal cohort, GRAND CANYON, in 4Q 2026. If data are positive, the Company will advance sevasemten toward a marketing application to seek approval as the first targeted therapy for this underserved population.
About Becker muscular dystrophy
Becker is a rare, genetic, X-linked neuromuscular disorder that predominantly affects males. Mutations in the dystrophin gene result in contraction-induced muscle damage, leading to progressive, irreversible muscle loss, motor function decline, and shortened lifespan. Symptoms can begin at any age, with ongoing functional deterioration throughout life. No approved therapies currently exist to treat the underlying disease.
About Sevasemten and the MESA Study
Sevasemten is an investigational orally administered therapy selectively modulating fast skeletal muscle contraction to reduce damage in muscular dystrophies. Sevasemten presents a novel mechanism of action designed to selectively limit the exaggerated muscle damage caused by the absence or loss of functional dystrophin. Sevasemten is being studied in late-stage clinical trials in Becker and Duchenne muscular dystrophy.
MESA is an open-label extension evaluating sevasemten long-term safety, tolerability, and efficacy in adults and adolescents with Becker previously treated in sevasemten clinical trials. For more information, see the MDA 2026 MESA poster here and clinicaltrials.gov (NCT06066580).
About Edgewise Therapeutics
Edgewise Therapeutics is a leading muscle disease biopharmaceutical company developing novel therapeutics for muscular dystrophies and serious cardiac conditions. The Company's deep expertise in muscle physiology is driving a new generation of novel therapeutics. Sevasemten is an orally administered first-in-class fast skeletal myosin inhibitor in late-stage clinical trials in Becker and Duchenne muscular dystrophies. EDG-7500 is a novel cardiac sarcomere modulator for the treatment of symptomatic hypertrophic cardiomyopathy, currently in Phase 2 clinical development. EDG-15400 is a novel cardiac sarcomere modulator for the treatment of heart failure, currently in Phase 1 clinical development. The entire team at Edgewise is dedicated to our mission: changing the lives of patients and families affected by serious muscle diseases. To learn more, go to edgewisetx.com or follow us on LinkedIn, X, Facebook and Instagram.
References
- Bello L, et al. Sci Rep. 2016;6:32439.
- Bello L, et al. Neurol Genet. 2025;11(4)e200285.
- De Wel B, et al. Eur J Neurol. 2024; 31(7):e16282.
- Schrama EJ, et al. Neurology. 2025;105(7):e214071.
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements as that term is defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Statements in this press release that are not purely historical are forward-looking statements. Such forward-looking statements include, among other things, statements regarding the potential of, and expectations regarding sevasemten, statements regarding Edgewise's expectations and milestones relating to its clinical trials and clinical development of sevasemten, including the timing of the readout of the GRAND CANYON pivotal cohort, statements regarding the potential outcome of the GRAND CANYON trial, statements regarding Edgewise's ability to obtain regulatory approval for sevasemten, statements regarding the potential for sevasemten to be the first-ever approved therapy for Becker, and statements by Edgewise's Chief Medical Officer. Words such as "believes," "anticipates," "plans," "expects," "intends," "will," "goal," "potential" and similar expressions are intended to identify forward-looking statements. The forward-looking statements contained herein are based upon Edgewise's current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those projected in any forward-looking statements due to numerous risks and uncertainties, including but not limited to: risks associated with Edgewise's limited operating history, its products being early in development and not having products approved for commercial sale; risks associated with Edgewise not having generated any revenue to date; Edgewise's ability to achieve objectives relating to the discovery, development and commercialization of its product candidates, if approved; Edgewise's need for substantial additional capital to finance its operations; Edgewise's substantial dependence on the success of sevasemten; Edgewise's ability to develop and commercialize sevasemten; risks related to Edgewise's clinical trials of its product candidates not demonstrating safety and efficacy; risks related to Edgewise's product candidates causing serious adverse events, toxicities or other undesirable side effects; the outcome of preclinical testing and early clinical trials not being predictive of the success of later clinical trials and the risks related to the results of Edgewise's clinical trials not satisfying the requirements of regulatory authorities; delays or difficulties in the enrollment and/or maintenance of patients in clinical trials; risks related to failure to capitalize on other indications or product candidates; risks related to competition; risks relating to interim, topline and preliminary data from Edgewise's clinical trials changing as more patient data becomes available; risks related to failure to develop a proprietary drug discovery platform; risks related to exposure to additional risk if we develop sevasemten in connection with other therapies; risks related to production of drugs by Edgewise's third-party manufacturers; risks related to changes in methods of product candidate manufacturing or formulation; risks related to not achieving adequate market acceptance; risks related to the patient population for our product candidates having a small patient population; risks related to the regulatory approval processes of domestic and foreign authorities being lengthy, time consuming and inherently unpredictable; risks relating to disruptions at the FDA, the SEC and other government agencies; risks relating to Edgewise's ability to attract and retain highly skilled executive officers and employees; Edgewise's ability to obtain and maintain intellectual property protection for its product candidates; Edgewise's reliance on third parties; risks related to future acquisitions or strategic partnerships; risks related to general economic and market conditions; and other risks. Information regarding the foregoing and additional risks may be found in the section entitled "Risk Factors" in documents that Edgewise files from time to time with the U.S. Securities and Exchange Commission. These forward-looking statements are made as of the date of this press release, and Edgewise assumes no obligation to update the forward-looking statements, or to update the reasons why actual results could differ from those projected in the forward-looking statements, except as required by law.
This press release contains hyperlinks to information that is not deemed to be incorporated by reference into this press release.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/edgewise-announces-positive-long-term-sevasemten-data-demonstrating-sustained-functional-stabilization-in-becker-muscular-dystrophy-through-3-5-years-of-follow-up-302708743.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/edgewise-announces-positive-long-term-sevasemten-data-demonstrating-sustained-functional-stabilization-in-becker-muscular-dystrophy-through-3-5-years-of-follow-up-302708743.html
SOURCE Edgewise Therapeutics